Textbook content produced by OpenStax is licensed under a Creative Commons Attribution License. Robinson, PhD, Klaus Theopold, Richard Langley We recommend using aĪuthors: Paul Flowers, Edward J. Use the information below to generate a citation. Earhart 2 of 2 All standard state, 25 ☌ and 1 bar (written to 1 decimal place). (Table III) there the accuracy was practically ideal. Standard absolute entropies at 1 bar and 298 K. 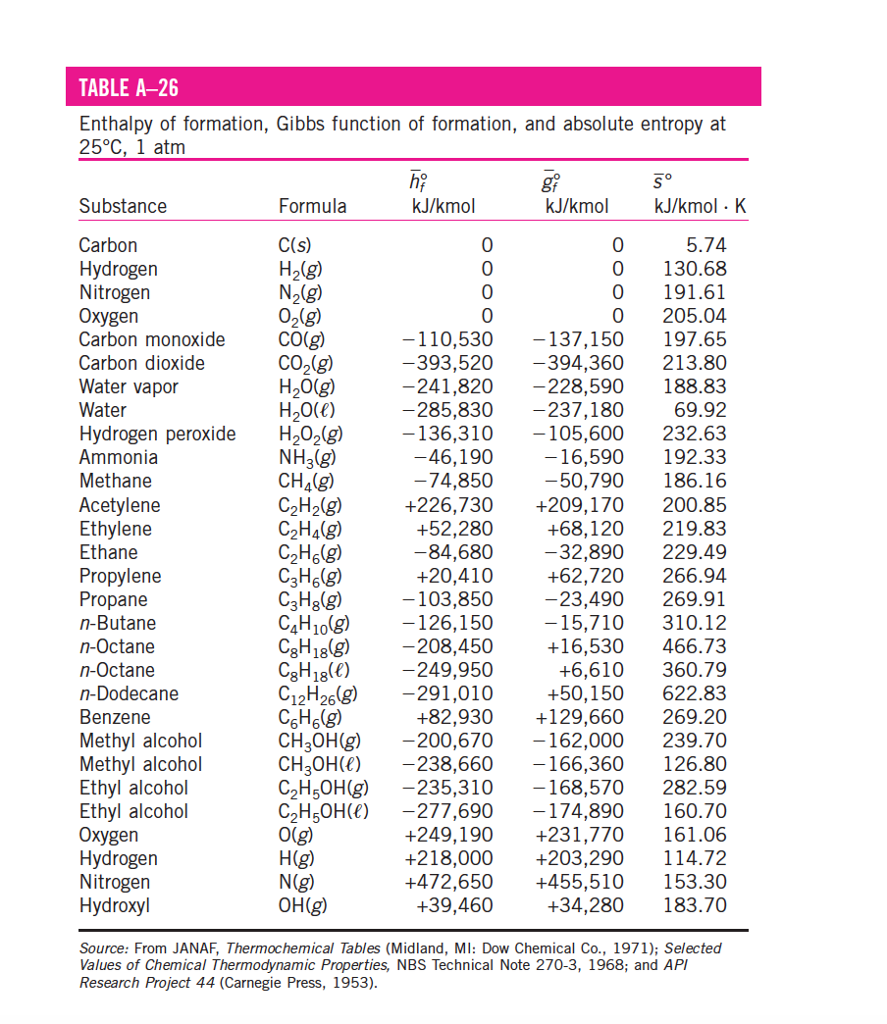
Some values at 1 bar and 298 K are given in Table 5.1. 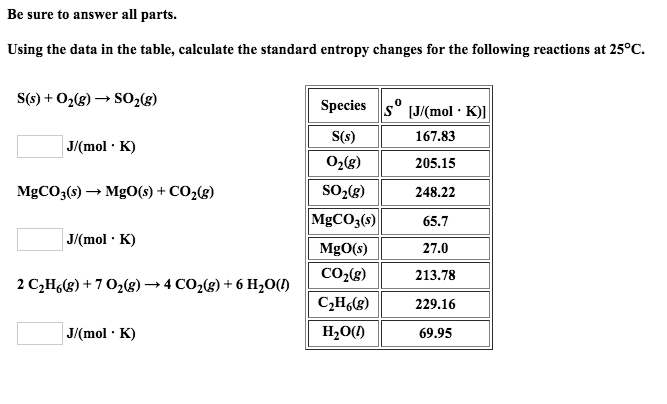
The example exercises that follow demonstrate the use of S° values in calculating standard entropy changes for physical and chemical processes. The data refer to the standard state at 1bar pressure and 298 K temperature. A partial listing of standard entropies is provided in Table 16.2, and additional values are provided in Appendix G. Then you must include on every digital page view the following attribution: From the change of K with temperature, standard enthalpy and entropy changes have been deter. The absolute entropy of elements and a large number of compounds have been obtained in this way and are listed in thermodynamic tables. If you are redistributing all or part of this book in a digital format, Then you must include on every physical page the following attribution: For any chemical reaction, the standard entropy change is the sum of the standard molar entropies of the products minus the sum of the standard molar entropies of the reactants. If you are redistributing all or part of this book in a print format, The standard molar entropy of a substance is the absolute entropy of 1 mole of the substance in the standard state. Want to cite, share, or modify this book? This book uses the This book may not be used in the training of large language models or otherwise be ingested into large language models or generative AI offerings without OpenStax's permission. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
